Sponsored Content
For over 50 years, dental unit waterline (DUWL) bacterial contamination has been a known hazard. Multiple cases of infection have been linked directly to waterlines over the past 20 years, however, it wasn’t until the large infection outbreaks in 2015 and 2016 at clinics in Atlanta and Anaheim involving over 90 children hit national headlines that the industry took a hard look at the risks.
Today, most practices have wisely taken the guidance of the 2003 Centers for Disease Control (CDC) Recommendations on Dental Water Quality and begun implementing antimicrobial treatment solutions designed specifically for DUWL maintenance.
Unfortunately, new data shows practices have to deal with a much higher fail rate among all treatment solutions than is acceptable.
RESEARCH DATA FROM OVER 22,000 CONSECUTIVE WATER TESTS
In 2017, ProEdge Dental Water Labs collected the largest dataset ever on the real-world effectiveness of waterline treatment solutions. Originally cited in “Treating and Monitoring Dental Water” by Nancy Dewhirst, RDH, BS and John A. Molinari, PhD which was published in Compendium’s E-Book Series, practices across the United States contributed their maintenance protocol information in 22,196 consecutive waterline tests.

Figure 1: R2A Agar Testing Pass/Fail Rates 2017 ©ProEdge Dental Water Labs
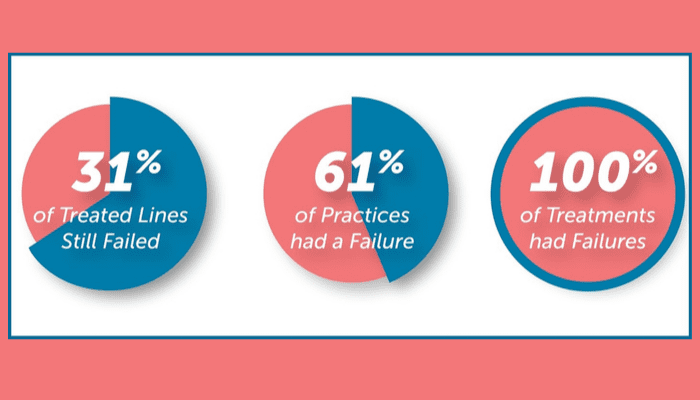
Three main takeaways stick out from the data:
- 31% of treated dental unit waterlines still failed to meet the CDC standard.
- 61% of practices had at least one failed waterline.
- 100% of treatment products had failures – categories fail rates ranged from 23% to 42%.

Figure 2: Research Takeaways ©ProEdge Dental Water Labs
See how your treatment product category
performed – download the data guide.
IMPLICATIONS YOUR ORGANIZATION NEEDS TO CONSIDER
Organizations and practices are making large investments in centralized purification/filtration systems, shock products, and continuous treatments like tablets or straws. With that level of investment and patient safety at stake, the expectations of performance are very high.
First, consider the data showed that every type of treatment solution – and each specific product – was represented in the failure data. No treatment solution proved to be a one-and-done silver bullet. The most popular treatment types, tablets and straws, failed at 23% and 27% respectively. Centralized systems had a 42% failure rate.
Of course, all treatment solutions can and do work. But the uniqueness of each practice’s waterline environment from temperature to usage to source water hardness may require you to tinker with the maintenance protocol to find the one that works best for your organization.
Second, pairing a routine shock treatment with a low-level antimicrobial (like a tablet or straw) showed the best performance. This aligns with the nature of chemical treatment and biofilm – shock products are strong enough to clean waterlines, and low-level antimicrobials are strong enough to inhibit attachment and abate growth. Eventually, low-level antimicrobials will be overrun and another shock will be necessary.
Think of it like the combination of brushing and professional dental cleaning. Brushing is vital, but that doesn’t mean a patient can stop going to the dentist. Both a cleaning and regular maintenance are necessary for effective biofilm control on teeth and the same is true for your waterlines.
And lastly, each practice needs to routinely monitor the effectiveness of their waterline maintenance protocol. If not, there is no way to know if your patients are safe and your practice is protected.
YOUR PRACTICE CAN ENSURE EFFECTIVE WATERLINE TREATMENT
To ensure your practice is making a good DUWL treatment investment, test your waterlines regularly. The CDC’s 2003 guidelines suggest following your dental unit manufacturer and waterline treatment product manufacturer’s instructions for testing frequency. However, many manufacturers have yet to make formal testing recommendations.
In the absence of direction from these manufacturers, the Organization for Safety, Asepsis, and Prevention recommends testing a minimum of quarterly.
“OSAP recommends that monitoring be performed periodically regardless of the product or protocol used to manage dental procedural water quality, even when manufacturer directions for monitoring are absent or unclear (p.7).”
“When there are no manufacturer directions available for dental units (e.g., older equipment) OSAP recommends that periodic monitoring and inspection should be performed according to the directions for use provided by the treatment product manufacturer or at least monthly on each dental unit or device… If monitoring results indicate water quality is acceptable for two consecutive monthly cycles, the frequency of testing may be reduced, but should not be less than every three months (p. 11).”
Documentation of routine waterline tests proving CDC-compliant water is not only a practice’s assurance their treatment investment is deserving, but also protects the organization should there be a complaint, inspection, or infection.
HERE’S WHAT YOU CAN DO NOW
I doubt I’m going out on a limb to say you probably didn’t get into dentistry to spend all your time becoming an expert on cleaning waterlines.
But our team at ProEdge Dental Water Labs did.
The team at ProEdge loves working with organizations searching for a data-backed answer to their waterline problem. If you’d like the help of an expert partner, ProEdge Dental Water Labs offers complimentary protocol consultations as well as free trials of its in-office and laboratory testing solutions.
www.ProEdgeDental.com | 888.843.3343 | support@proedgedental.com
If you’d like to learn more about these opportunities,
contact Kelley Birschbach at KelleyB@ProEdgeDental.com to get started.
Read the Legal Implications of Dental Water Non-Compliance & 3 Ways to Start Protecting Your Organization article.
Looking for a Job? Looking to Fill a Job? JoinDSO.com can help: Subscribe for free to the most-read and respected
Subscribe for free to the most-read and respected
resource for DSO analysis, news & events: Read what our subscribers & advertisers think of us:
Read what our subscribers & advertisers think of us: